 In the gas phase collisions are rare compared to those of solvent in the condensed phase and so vibrational relaxation can no longer compete with fluorescence and so the fluorescence spectrum can arise from a mixture of vibrational levels not just v'=0. In the question the molecule also shows a mirror image but the picture is in wavelength not wavenumber (or energy) so is a bit distorted. The difference between Emission and Absorption Spectra is self-explanatory where Absorption Spectrum constitutes the missing incident frequencies of radiation which are absorbed by the sample material when its electrons transition from a low energy state to a high energy state. Test 1 2 3 4 5 6 Line emission and absorption spectra Studying the line spectra produced by hot gases and absorbed by cooler gases allows us to identify the elements in stars. Emission Spectrum: Starlight can also heat up a cloud of gas, exciting the atoms and molecules within the gas, and causing it to emit light. Assuming that the ground and excited state have a similar shape the fluoresce and absorption spectra are mirror images of one another as shown in the figure. An absorption spectrum has dark lines or gaps in the spectrum corresponding to wavelengths that are absorbed by the gas. The wavelengths of a particular element’s absorption lines are the same as the wavelengths of its emission lines. The absorption and emission spectra of each element are inverses of each other. Every element has a unique set of absorption and emission lines, or spectral signature.  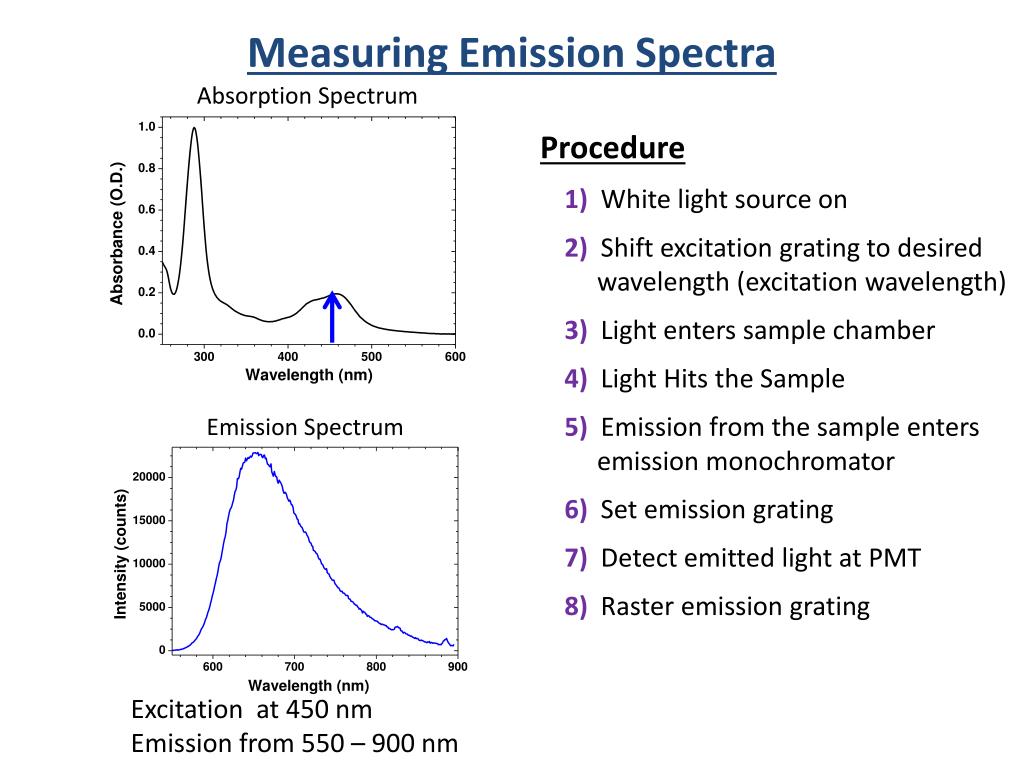
The amount of energy it loses will be equal to the difference in. Simplified illustration of absorption and emission spectra. Thus all the excited molecules are now in v'=0 in the excited state. If an electron is in an excited state it can return to a lower energy level. The intensity of absorption/emission is proportional to the transition moment squared, $d_$. When the species come back to the ground state from the excited state. When an atom or molecule excites, it absorbs a certain energy in the electromagnetic radiation therefore, that wavelength will be absent in the recorded absorption spectrum. Swenberg, Electronic Processes in Organic Crystals and Polymers, 1999)Īssuming harmonic oscillations, the overlap between each two vibrational wavefunctions (different energy level for different frequencies/number of nodes) is defined as the nuclear vibrational overlap, which in turn contributes to the total transition moment. Difference between emission and absoroption spectrum. When an electron is excited from a ground state $S_0$ to the first excited state $S_1$, the bond is stretched and the internuclear separation increases: 
As mentioned, the reason for this difference is the contribution of nuclear vibrational overlap to the transition moment. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
